Yena Lee

AI initiatives often get stuck in the Proof of Concept (POC) stage or fail to deliver tangible business value. The team might "build the solution," but it doesn’t address the real problem.

Enterprises often struggle with rigid, outdated front-end systems that drive up costs and hinder their ability to adapt to market demands. Here's what a modern, AI-ready tech stack looks like and how you can achieve AI readiness incrementally in complex enterprise environments.
Learn how to launch promotional banners and sales content seamlessly across your site without developer bottlenecks. We cover how to audit your sales content workflows to create a headless e-commerce roadmap that works for your brand and walk through examples of how we’ve solved sales campaigns for major e-commerce brands.

A practical guide to using AI to curate product collections at scale, make product recommendation and search algorithms smarter with semantic awareness, and empower teams to deliver seamless customer experiences.

Generate nuanced translations for your e-commerce site using AI. Learn how to set up, fine-tune, and integrate AI models seamlessly with your headless CMS for automatic content translation. Uncover practical steps for implementation, from choosing the right language model to architecting efficient translation workflows that align with your budget and goals.
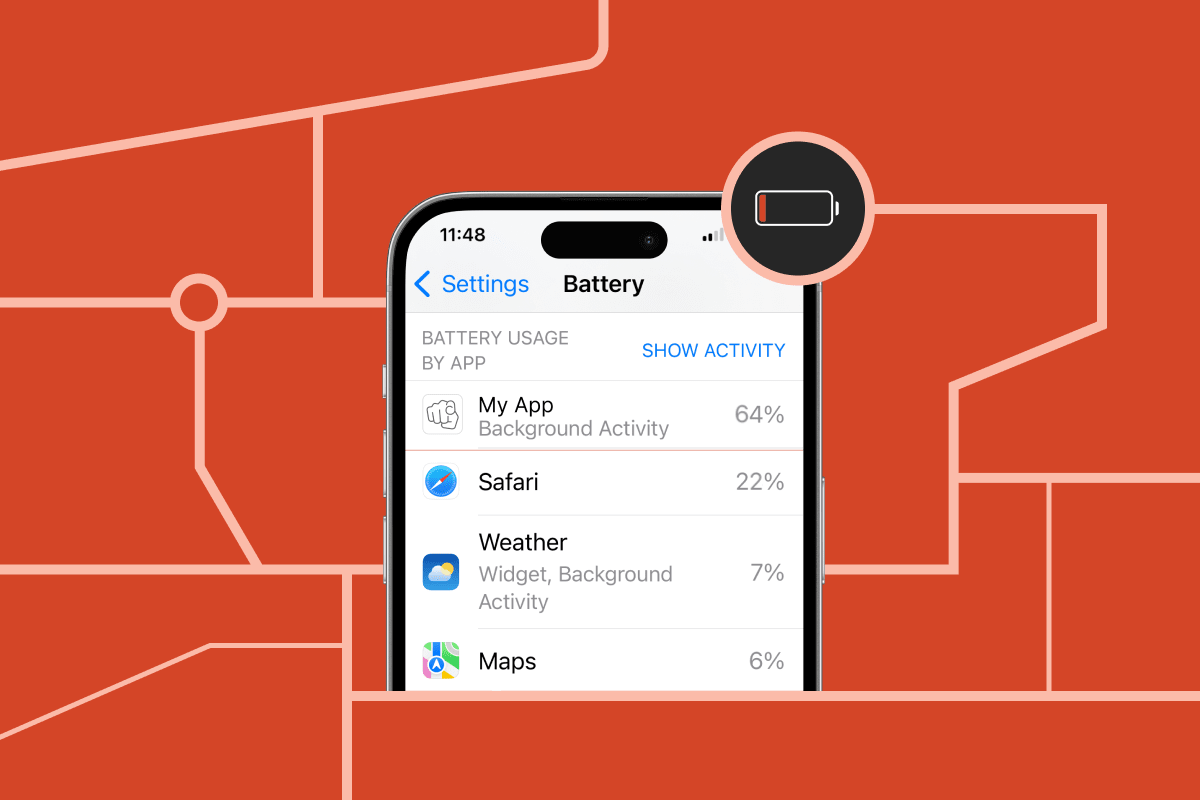
Lowering location accuracy is not the best way to reduce your app's power usage. Use these configurations for iOS background location services and stop draining your users' batteries.
Choosing a headless CMS can be a daunting task. We created a tool for you to chat with our headless platform expert Scott Fuller to help you find the best headless solution for your business.
Contentful, Sanity, and Strapi are the headless CMS platforms we recommend most often to our clients. Here's a comparison of features that matter, plus an in-depth guide to choosing the right headless CMS platform for you.







